Peter Lobner
I described the Qualcomm Tricorder XPrize competition in my 10 March 2015 post, “Medical Tricorder Technology is Closer Than you Think.” The goal of the competition is to develop a real-world equivalent of the Star Trek Tricorder, with the following basic capabilities and features:
- Diagnose at least 13 different health conditions including the following nine required conditions: anemia, atrial fibrillation, chronic obstructive pulmonary disease, diabetes, leukocytosis, pneumonia, ottis media, sleep apnea and urinary tract infection.
- Weigh less than five pounds
At the time of my last update in December 2015, the following seven teams had been selected to compete in the extended Final Round for $10 million in prize money.
- Aezon (U.S.)
- Clouddx (Canada)
- Danvantri (India)
- DMI (U.S.)
- Dynamical Biomarkers Group (Taiwan)
- Final Frontier Medical Devices (U.S.)
- Intellesens-Scanadu (UK)
Each of these teams submitted their final working prototypes for evaluation in Q3 2016. On 13 December 2016, Qualcomm Tricorder XPrize announced that they had selected two teams to continue into the finals:
“Congratulations to our two final teams, Dynamical Biomarkers Group and Final Frontier Medical Devices, who will proceed to the final phase in the $10M Qualcomm Tricorder XPRIZE. Both teams’ devices will undergo consumer testing over the next few months at the Altman Clinical Translational Research Institute at the University of California San Diego, and the winner will be announced in Q2, 2017.”
Both teams are required to deliver 45 kits for testing.
The XPrize will be split with $6 million going to the winning team, $2 million going to the runner-up, and $1 million for the team that receives the highest vital signs score in the final round. An additional $1M already has been awarded in milestone prizes.
The two competing devices are briefly described below. For more information, visit the Qualcomm Tricorder XPrize website at the following link:
Dynamical Biomarkers Group
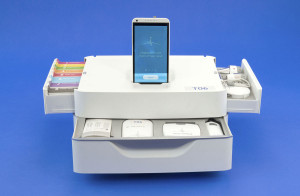 Source: Qualcomm Tricorder XPrize
Source: Qualcomm Tricorder XPrize
Key system features:
- Comprised of three modules: Smart Vital-Sense Monitor; Smart Blood-Urine Test Kit; Smart Scope Module.
- Includes technologies for physiologic signal analysis, image processing, and biomarker detection.
- Smartphone app executes simple, interactive screening process that guides the users to carry out specific tests to generate disease diagnosis. The phone’s on-board camera is used to capture images of test strips. The smartphone communicates to the base unit via Bluetooth.
- The base unit uploads collected data to a remote server for analysis.
Final Frontier Medical Devices: DxtER
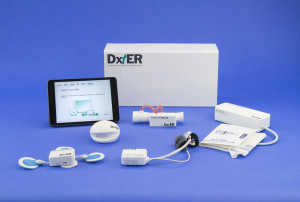 Source: Qualcomm Tricorder XPrize
Source: Qualcomm Tricorder XPrize
Key system features:
- DxtER is designed as a consumer product for monitoring your health and diagnosing illnesses in the comfort of your own home.
- Non-invasive sensors collect data about your vital signs, body chemistry, and biological functions.
- An iPad Mini with an on-board AI diagnostic app synthesizes the health data to generate a diagnosis.
- While DxtER functions autonomously, it also can share data with a remote healthcare provider.
Best wishes to both teams as they enter the final round of this challenging competition, which could significantly change the way some basic medical services are delivered in the U.S. and around the world.