Peter Lobner
In my 19 December 2016 post, “What to do with Carbon Dioxide,” I provided an overview of the following three technologies being developed for underground storage (sequestration) or industrial utilization of carbon dioxide:
- Store in basalt formations by making carbonate rock
- Store in fractures in deep crystalline rock
- Make ethanol
You can read this post here:
https://lynceans.org/all-posts/what-to-do-with-carbon-dioxide/
In the past two years, significant progress has been made in the development of processes to convert gaseous carbon dioxide waste streams into useful products. This post is intended to highlight some of the advances being made and provide links to additional current sources of information on this subject.
1. Carbon XPrize: Transforming carbon dioxide into valuable products
The NRG / Cosia XPrize is a $20 million global competition to develop breakthrough technologies that will convert carbon dioxide emissions from large point sources like power plants and industrial facilities into valuable products such as building materials, alternative fuels and other items used every day. You’ll find details on this competition on the XPrize website at the following link:
https://www.xprize.org/prizes/carbon
As shown in the following timeline from the above website, this competition started in September 2015.
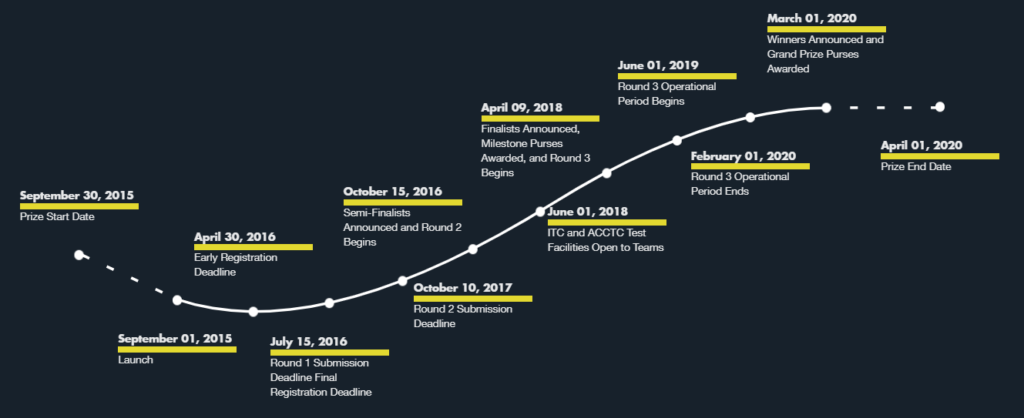
In April 2018, the following ten international teams were selected as finalists:
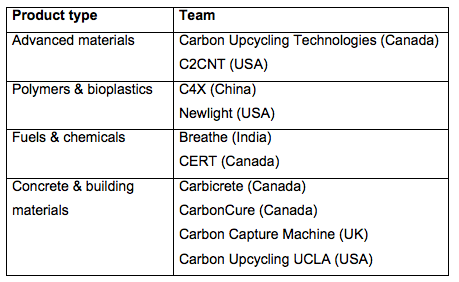
- Four teams from Canada: Carbicrete, Carbon Upcycling Technologies, CarbonCure and CERT
- Three teams from USA: C2CNT, Carbon Upcycling UCLA and Newlight
- One team from UK: Carbon Capture Machine
- One team from India: Breathe
- One team from China: C4X
The processes being developed by these teams will produce a range of valuable products:
You can read more about each of the competing teams and their intended products in the XPrize finalist selection report here:
The competition is now in the testing and certification phase. Each team is expected to scale up their pilot systems by a factor of 10 for the operational phase, which starts in June 2019 at the Wyoming Integrated Test Center and the Alberta (Canada) Carbon Conversion Technology Center.
The teams will be judged by the amount of carbon dioxide converted into usable products and the value of those products. We’ll have to wait until the spring of 2020 for the results of this competition.
2. World’s largest post-combustion carbon capture project
Post-combustion carbon capture refers to capturing carbon dioxide from flue gas after a fossil fuel (e.g., coal, natural gas or oil) has been burned and before the flue gas is exhausted to the atmosphere. You’ll find a 2016 review of post-combustion carbon capture technologies in the paper by Y. Wang, et al., “A Review of Post-combustion Carbon Dioxide Capture Technologies from Coal-fired Power Plants,” which is available on the ScienceDirect website here:
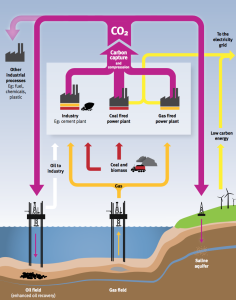
In January 2017, NRG Energy reported the completion of the Petra Nova post-combustion carbon capture project, which is designed to remove 90% of the carbon dioxide from a 240 MW “slipstream” of flue gas at the existing W. A. Parish generating plant Unit 8. The “slipstream” represents 40% of the total flue gas flow from the coal-fired 610 MW Unit 8. To date, this is the largest post-combustion carbon capture project in the world. Approximately 1.4 million metric tons of carbon dioxide will be captured annually using a process jointly developed by Mitsubishi Heavy Industries, Ltd. (MHI) and the Kansai Electric Power Co. The US Department of Energy (DOE) supported this project with a $190 million grant.
The DOE reported: “The project will utilize a proven carbon capture process, which uses a high-performance solvent for carbon dioxide absorption and desorption. The captured carbon dioxide will be compressed and transported through an 80 mile pipeline to an operating oil field where it will be utilized for enhanced oil recovery (EOR) and ultimately sequestered (in the ground).”
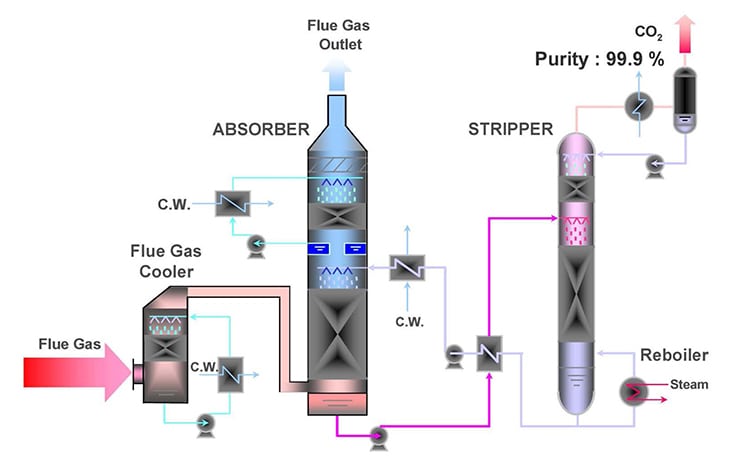
Source: National Energy Technology Laboratory


Source: Business Wire
You’ll find more information on the Petra Nova project at the following links:
- Aaron Larson, “World’s Largest Post-Combustion Carbon Capture Project Completed,” PowerMag.com, 10 January 2017: https://www.powermag.com/worlds-largest-post-combustion-carbon-capture-project-completed/
- “Petra Nova – W.A. Parish Project,” USDOE, Office of Fossil Energy: https://www.energy.gov/fe/petra-nova-wa-parish-project
- Petra Nova is one of two carbon capture and sequestration power plants in the world, EIA, 31 October 2017: https://www.eia.gov/todayinenergy/detail.php?id=33552#
3. Pilot-scale projects to convert carbon dioxide to synthetic fuel
Thyssenkrupp pilot project for conversion of steel mill gases into methanol
In September 2018, Thyssenkrupp reported that it had “commenced production of the synthetic fuel methanol from steel mill gases. It is the first time anywhere in the world that gases from steel production – including the carbon dioxide they contain – are being converted into chemicals. The start-up was part of the Carbon2Chem project, which is being funded to the tune of around 60 million euros by Germany’s Federal Ministry of Education and Research (BMBF)……..‘Today the Carbon2Chem concept is proving its value in practice,’ said Guido Kerkhoff, CEO of Thyssenkrupp. ‘Our vision of virtually carbon dioxide-free steel production is taking shape.’”
You can read the Thyssenkrupp press release here:
https://www.thyssenkrupp.com/en/newsroom/press-releases/press-release-141984.html
Berkeley Laboratory developing a copper catalyst that yields high efficiency carbon dioxide-to-fuels conversion
The DOE Lawrence Berkeley National Laboratory (Berkeley Lab) has been engaged for many years in creating clean chemical manufacturing processes that can put carbon dioxide to good use. In September 2017, Berkeley Lab announced that its scientists has developed a new electrocatalyst comprised of copper nanoparticles that can directly convert carbon dioxide into multi-carbon fuels and alcohols (e.g., ethylene, ethanol, and propanol) using record-low inputs of energy. For more information, see the Global Energy World article here:
4. Pilot-scale “Negative Emissions Technology” plants
The term negative emissions technology (NET) refers to an industrial processes designed to remove and sequester carbon dioxide directly from the ambient atmosphere rather than from a large point source of carbon dioxide generation (e.g. the flue gas from a fossil-fueled power generating station or a steel mill). Think of a NET facility as a carbon dioxide removal “factory” that can be sited independently from the sources of carbon dioxide generation.
The Swiss firm Climeworks is in the business of developing carbon dioxide removal factories using the following process:
“Our plants capture atmospheric carbon with a filter. Air is drawn into the plant and the carbon dioxide within the air is chemically bound to the filter. Once the filter is saturated with carbon dioxide it is heated (using mainly low-grade heat as an energy source) to around 100 °C (212 °F). The carbon dioxide is then released from the filter and collected as concentrated carbon dioxide gas to supply to customers or for negative emissions technologies. Carbon dioxide-free air is released back into the atmosphere. This continuous cycle is then ready to start again. The filter is reused many times and lasts for several thousand cycles.”
This process is shown in the following Climeworks diagram:
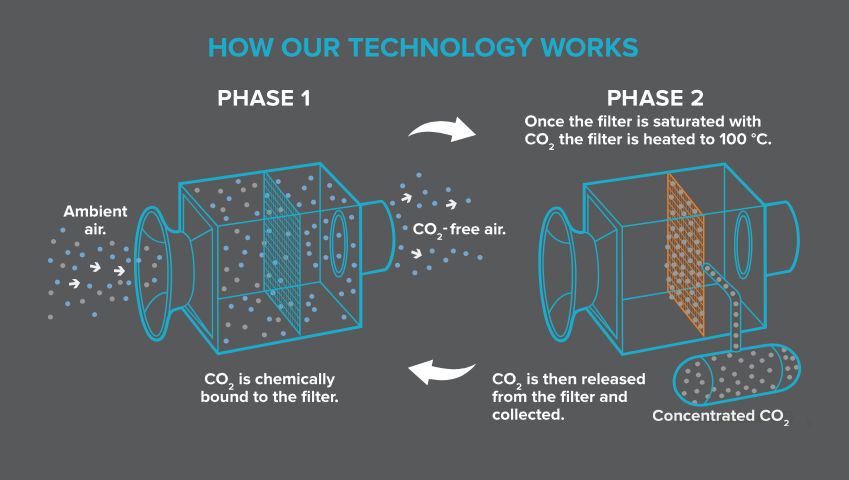
You’ll find more information on Climeworks on their website here:
Climeworks NET in Iceland
In 2017, Climeworks began operation in Iceland of their first pilot facility to remove carbon dioxide from ambient air and produce concentrated carbon dioxide that is injected into underground basaltic rock formations, where the carbon dioxide gets converted into carbonite minerals in a relatively short period of time (1 – 2 years) and remains fixed in the rock. Climeworks uses waste heat from a nearby geothermal generating plant to help run their carbon capture system. This process is shown in the following diagram.
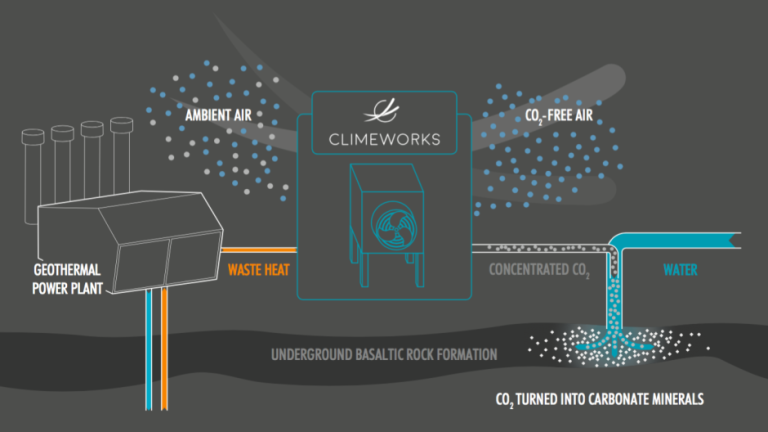
This small-scale pilot facility is capable of removing only about 50 tons of carbon dioxide from the atmosphere per year, but can be scaled up to a much larger facility. You’ll find more information on this Climeworks project here:
https://www.popularmechanics.com/science/green-tech/news/a28629/first-negative-emissions-plant/
Climeworks NET in Italy
In October 2018, Climeworks began operation in Italy of another pilot-scale NET facility designed to remove carbon dioxide from the atmosphere. This facility is designed to remove 150 tons of carbon dioxide from the atmosphere per year and produce a natural gas product stream from the atmospheric carbon dioxide, water, and electricity. You’ll find more information on this Climeworks project here:
https://www.popularmechanics.com/science/green-tech/a23621438/climeworks-co2-methane-facility/
5. Consensus reports on waste stream utilization and negative emissions technologies (NETs)
The National Academies Press (NAP) recently published a consensus study report entitled, “Gaseous Carbon Waste Streams Utilization, Status and Research Needs,” which examines the following processes:
- Mineral carbonation to produce construction material
- Chemical conversion of carbon dioxide into commodity chemicals and fuels
- Biological conversion (photosynthetic & non-photosynthetic) of carbon dioxide into commodity chemicals and fuels
- Methane and biogas waste utilization
The authors note that, “previous assessments have concluded that …… > 10 percent of the current global anthropogenic carbon dioxide emissions….could feasibly be utilized within the next several decades if certain technological advancements are achieved and if economic and political drivers are in place.”

You can download a free pdf copy of this report here:
https://www.nap.edu/catalog/25232/gaseous-carbon-waste-streams-utilization-status-and-research-needs
Also on the NAP website is a prepublication report entitled, “Negative Emissions Technologies and Reliable Sequestration.” The authors note that NETs “can have the same impact on the atmosphere and climate as preventing an equal amount of carbon dioxide from being emitted from a point source.”

You can download a free pdf copy of this report here:
In this report, the authors note that recent analyses found that deploying NETs may be less expensive and less disruptive than reducing some emissions at the source, such as a substantial portion of agricultural and land-use emissions and some transportation emissions. “ For example, NAPs could be a means for mitigating the methane generated from enteric fermentation in the digestive systems of very large numbers of ruminant animals (e.g., in the U.S., primarily beef and dairy cattle). For more information on this particular matter, please refer to my 31 December 2016 post, “Cow Farts Could be Subject to Regulation Under a New California Law,” which you’ll find here:
https://lynceans.org/all-posts/cow-farts-could-be-subject-to-regulation-under-a-new-california-law/







 The 1885 periodic table. Source: University of St. Andrews
The 1885 periodic table. Source: University of St. Andrews